
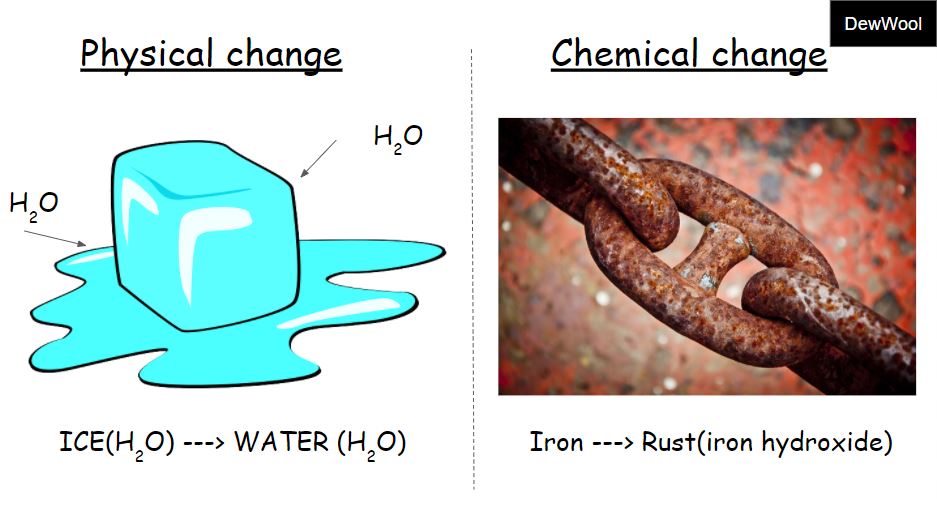
Note the properties of a mixture may be different from its components. Mixtures - Mixing together materials where one is not soluble in the other is a physical change.This is a physical change because it's not permanent and no chemical reaction occurs. Magnetism - If you hold a magnet up to iron, you'll temporarily magnetize it.Phase Changes - Altering the temperature and/or pressure can change the phase of a material, yet its composition is unchanged,.The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The wax molecules are undergoing a chemical change they are changing into different molecules by reacting with a substance in the air. The process of burning (as opposed to evaporating) is a chemical reaction, a chemical change. Tearing of paper, melting/freezing of water, cutting of trees, etc. Chemical Change is a process in which the substance transforms into a new substance, having different chemical composition. Physical change refers to a change in which the molecules are rearranged but their internal composition remains same. What are Class 6 physical and chemical changes? No new products are formed during the physical change of a matter. The physical change only affects the physical properties of the matter i.e size, shape, volume, density, state or colour. The change of matter which occurs without changing its chemical composition is called a physical change. What are the 20 examples of chemical changes? Is boiling water a physical change?ĭuring boiling, water changes from liquid to gaseous state. The five conditions of chemical change: color change, formation of a precipitate, formation of a gas, odor change, temperature change. What are 2 examples of a chemical change?Įxamples of chemical changes include baking soda and vinegar creating carbon dioxide, iron rusting, and wood burning. Biological decomposition or fermentation.Freezing of water is a physical change because only the physical state of water is changed i.e. What are various types of changes?Īnswer: In a physical change, only the physical properties of a substance change and no new substances are formed. People’s initial reaction to the change will likely be shock or denial as they refuse to accept that change is happening. The stages are shock, anger, acceptance and commitment. ☛There are two types of change in matter: physical change and chemical change. Many physical changes are reversible, if sufficient energy is supplied. Examples of physical changes are boiling, melting, freezing, and shredding. What are 5 physical and chemical changes?Įxamples of chemical changes are burning, cooking, rusting, and rotting. 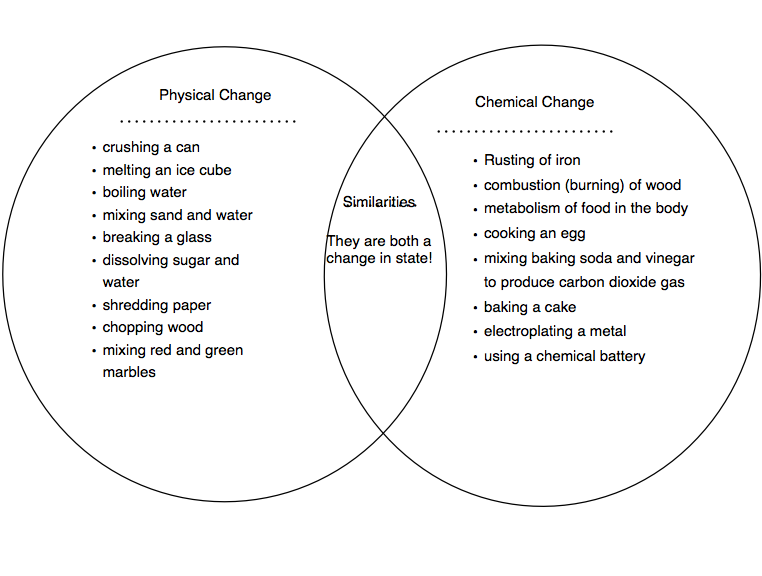
Cutting, bending, dissolving, freezing, boiling, and melting are some of the processes that create physical changes. Physical changes include transitions from one state to another, such as from solid to liquid or liquid to gas.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
